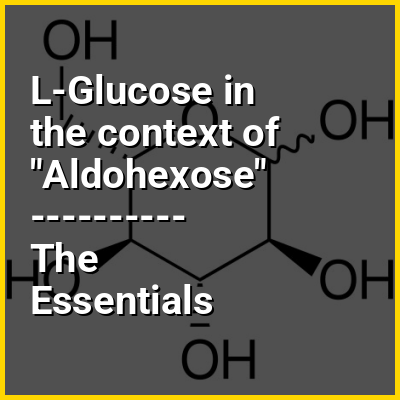
l-Glucose is an organic compound with formula C6H12O6 or O=CH[CH(OH)]5H, specifically one of the aldohexose monosaccharides. As the l-isomer of glucose, it is the enantiomer of the more common d-glucose.
l-Glucose does not occur naturally in living organisms, but can be synthesized in the laboratory. l-Glucose is indistinguishable in taste from d-glucose, but cannot be used by living organisms as a source of energy because it cannot be phosphorylated by hexokinase, the first enzyme in the glycolysis pathway. One of the known exceptions is in Trinickia caryophylli, a plant pathogenic bacterium, which contains the enzyme d-threo-aldose 1-dehydrogenase which is capable of oxidizing l-glucose.